Iron disulfide ( FeS2 ) is a natural earth-abundant and nontoxic material with possible applications in lithium batteries, transistors or photovoltaic (PV) devices. According to the analysis carried out by Wadia et al., among 23 semiconducting materials, FeS2 is the best candidate for the development of large-scale solar cells at low cost (<2 × 10−6 ¢/W). Furthermore, FeS2 exhibits excellent optoelectronic properties such as a band gap of 0.8 to 1.38 eV, a high optical absorption coefficient (2 × 105 cm−1), high carrier mobility (2 to 80 cm2/Vs) and a large charge carrier lifetime (200 ps). Therefore, FeS2 nanoparticles (NPs) can be a good alternative for PV applications.*
In “Nontoxic pyrite iron sulfide nanocrystals as second electron acceptor in PTB7:PC71BM-based organic photovoltaic cells “ Olivia Amargós-Reyes, José-Luis Maldonado, Omar Martínez-Alvarez, María-Elena Nicho, José Santos-Cruz, Juan Nicasio-Collazo, Irving Caballero-Quintana and Concepción Arenas-Arrocena report the synthesis of nontoxic pyrite iron sulfide ( FeS2 ) nanocrystals (NCs) using a two-pot method. Moreover, they study the influence of these NCs incorporated into the PTB7:PC71BM active layer of bulk-heterojunction ternary organic photovoltaic ( OPV ) cells.*
The AFM roughness images presented in this article were acquired in dynamic force mode using NANOSENSORS™ PointProbe® Plus PPP-NCLAu AFM probes.
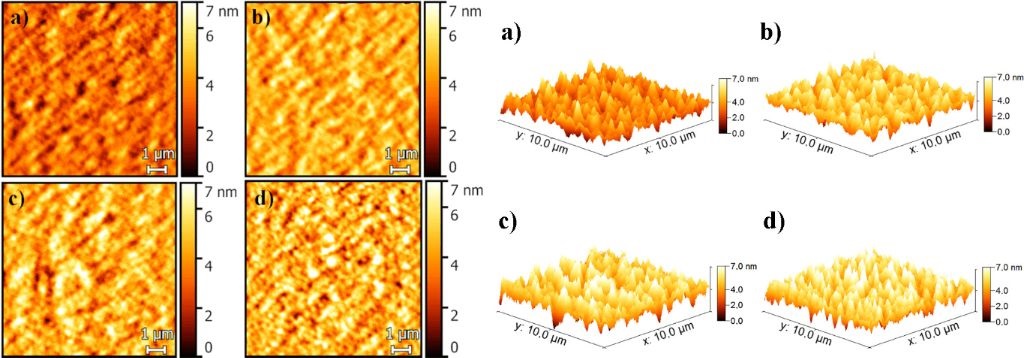
2D (left) and 3D (right) AFM images of the OPVs with different concentrations of FeS2
(a) 0.0 wt %, b) 0.25 wt %, c) 0.5 wt % and d) 1.0 wt %) recorded in noncontact mode.
*Olivia Amargós-Reyes, José-Luis Maldonado,
Omar Martínez-Alvarez, María-Elena Nicho, José Santos-Cruz, Juan
Nicasio-Collazo, Irving Caballero-Quintana and Concepción Arenas-Arrocena
Nontoxic pyrite iron sulfide nanocrystals as second electron acceptor in PTB7:PC71BM-based
organic photovoltaic cells
Beilstein Journal of . Nanotechnology 2019, 10, 2238–2250.
DOI: doi:10.3762/bjnano.10.216
Please follow this external link to read the full article: https://www.beilstein-journals.org/bjnano/articles/10/216#R65
Open Access: The article “Nontoxic pyrite iron sulfide nanocrystals as second electron acceptor in PTB7:PC71BM-based organic photovoltaic cells” by Olivia Amargós-Reyes, José-Luis Maldonado, Omar Martínez-Alvarez, María-Elena Nicho, José Santos-Cruz, Juan Nicasio-Collazo, Irving Caballero-Quintana and Concepción Arenas-Arrocena is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.